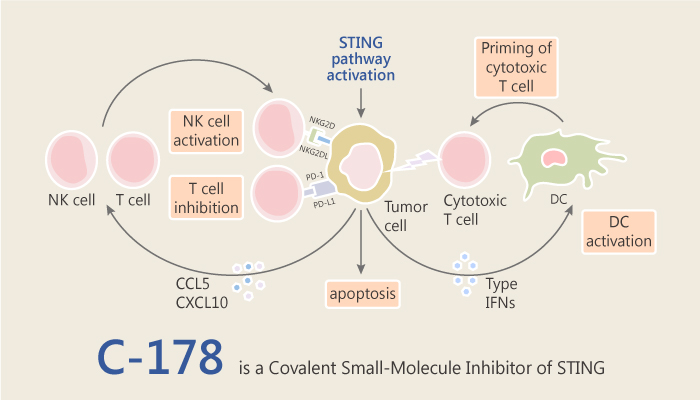
In the previous blog, I have introduced C-176 as a strong and covalent mouse STING inhibitor. Today, I’d like to introduce another STINF inhibitor C-178 in the same article. STING, short for stimulator of interferon genes, also known as transmembrane protein 173 and plays an important role in innate immunity by inducing type I interferon production.
In the study, C-178 potently and selectively suppressed the STING responses elicited by distinct bona fide activators. In addition, C-178 inhibited the CMA-induced phosphorylation of TBK1. TBK1 is a key downstream protein kinase of STING. Notably, however, C-178 did not appreciably affect STING responses in human cells. The species-specific activity of C-178 and C-176 suggested that the compounds directly target mouse STING (mmSTING) but not human STING (hsSTING).
C-178, however, neither impaired the trafficking of STING to the Golgi apparatus nor affected the endolysosomal degradation of STING thereafter. The authors then suggested that C-178 may alter post-translational modifications of STING. They focused on palmitoylation because this had previously been reported to rely on Cys91 and to be involved in the activation of TBK126. Indeed, they found that CMA-induced palmitoylation of STING was markedly attenuated in the presence of C-178. However, the palmitoylation of the transferrin receptor or calnexin had no apparent changes.
C-178 is a covalent inhibitor of STING, binding to Cys91. Additionally, it potently and selectively suppresses the STING responses elicited by distinct bona fide activators in mouse but not human. C-178 significantly reduces STING-, but not RIG-I- or TBK1-, mediated IFN-β reporter activity. C-178 blocks palmitoylation (PMA)-induced clustering of STING; inhibits the CMA-induced phosphorylation of TBK1 (downstream protein kinase of STING).
To sum up, the innovative study revealed the inhibitory mechanism of STING on pharmacology and showed the potential of STING inhibitors in the treatment of autoinflammatory diseases.