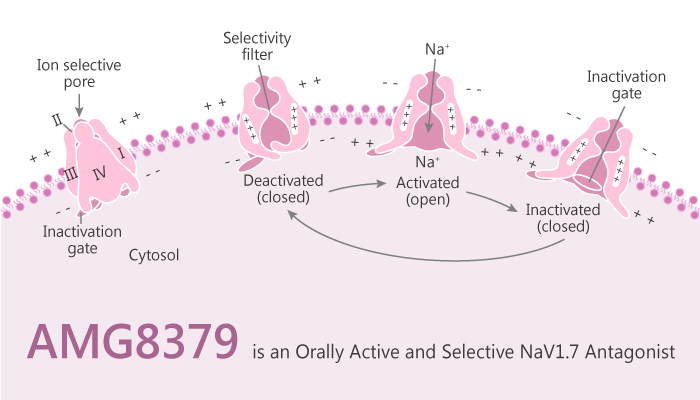
Potent and selective antagonists of the voltage-gated sodium channel Nav1.7 can be a potential method for the development of chronic pain therapies. In this article, we will introduce an atropisomer quinolone sulfonamide antagonist AMG8379.
In a whole-cell patch-clamp electrophysiology assay. AMG8379 can inhibit human Nav1.7 channels with an IC50 of 8.5 nM and endogenous tetrodotoxin (TTX)-sensitive sodium channels in dorsal root ganglion (DRG) neurons with an IC50 of 3.1 nM.
AMG8379 exhibits 100-1000 selective over Nav1.4, Nav1.5 and TTX-resistant NaV channels in DRG neurons. The IC50 for AMG8379 inhibition of C-fiber spiking based on the level of firing in Nav1.7 KO mice representing a complete pharmacological block of the Nav1.7-component of this assay is calculated. In this manner, the IC50 for AMG8379 block is 47.0 nM.
As a result, UVB irradiation to the plantar surface of the hind paw can lead to thermal hyperalgesia in mice. The anti-inflammatory agent naproxen completely reverses thermal hypersensitivity. Besides, the mean withdrawal latency returning to the level like the nonirradiated-contralateral paw.
When AMG8379 at low doses, this agent does not impose too much affect on UVB-induced thermal hyperalgesia. However, AMG8379 at a dose of 100 mg/kg can cause to an analgesic effect similar to naproxen. Besides the paw withdrawal latency of the ipsilateral side has no difference between the contralateral side.
TRPV1 agonist capsaicin produces a strong nociceptive response in mice. Observed mice often lift and lick their hind paw over the 5-minute observation period. AMG8379 exhibits a dose-dependent reduction in nociceptive behavior. Also, it leads to a prohibitively obvious reduction in total time in paw licking.
In conclusion, the Nav1.7-selective and central nervous system engages and binds channels both on peripheral nerve fibers and in central terminals of primary afferent nociceptors.
Nav1.7-preferring small-molecule antagonists block spontaneous or evoked pain responses in vivo. And blockage of peripheral Nav1.7 channels can mitigate pain in humans. AMG8379, as a selective Nav1.7 channels antagonist of human and mouse in neurons. It inhibits C-fiber action potential firing in a mouse skin-nerve preparation in vitro. Meanwhile, AMG8379 blocks capsaicin-induced licking, and UVB-induced thermal hyperalgesia in vivo.
AMG8379 provides a potential for the further investigation of Nav1.7 channels antagonist s for pain relief.
Reference:
Kornecook TJ, et al. J Pharmacol Exp Ther. 2017 Jul;362(1):146-160.