CD40 (TNFRSF5) and CD40L (CD154, TNFSF5) are the tumor necrosis factor (TNF)-TNFR superfamily (TNFSF) costimulatory molecules, and this protein-protein interaction is among the most extensively studied TNFSF members. CD40 is a transmembrane glycoprotein constitutively expressed on antigen-presenting cells (APCs), whereas its ligand, CD40L, exists both as a transmembrane protein and a soluble form in the plasma. The interaction between CD40L on T cells and its receptor CD40 on B cells is essential for lymphocyte signaling leading to T cell-dependent B cell proliferation, immunoglobulin class switching, and B-cell maturation.
CD40–CD40L interaction plays an important role in glucolipotoxicity-induced β-cell death.
In this study, Jinshui Chen et al report the identification of DRI-C21045. Especially, DRI-C21045 is a potent inhibitor of the CD40-CD40L interaction. DRI-C21045 inhibits CD40, TNF, and NF-κB with IC50s of 0.17 μM, 992.0 μM, and 17.1 µM, respectively. Moreover, DRI-C21045 shows concentration-dependent inhibition of the activation of NF-κB in CD40 sensor cells. DRI-C21045 inhibits CD40L-induced activation in NF-κB sensor cells, THP-1 myeloid cells, and primary human B cells with IC50 in the low micromolar range. CD40L alone indeed induces increased expression. However, DRI-C21045 concentration-dependently inhibits this effect. DRI-C21045 also concentration-dependently inhibits CD40L-induced B cell proliferation in a statistically significant manner at concentrations higher than 10 µM. DRI-C21045 (20-60 mg/kg; s.c.; b.i.d.) significantly inhibits alloantigen-induced T cell response in a dose-responsive manner.
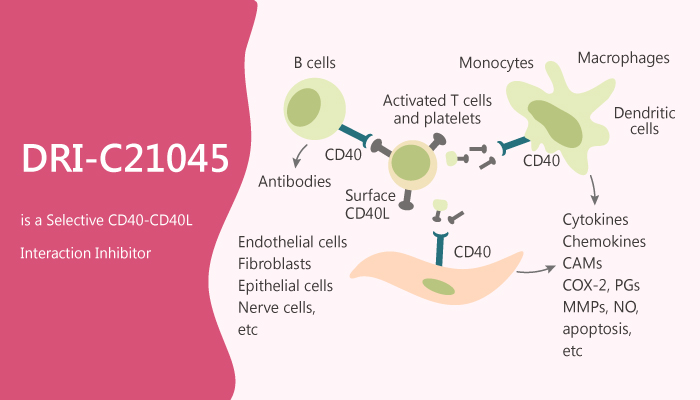
In summary, DRI-C21045 provides proof-of-principle evidence for the possibility of small-molecule inhibition of co-stimulatory protein-protein interactions, establish the structural requirements needed for efficient CD40-CD40L inhibition, and serve to guide the search for such immune therapeutics.