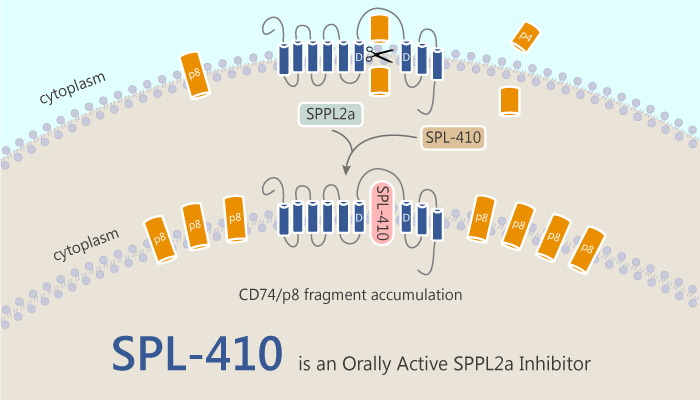
The immune system is a powerful host defense mechanism specializing in antigen recognition. Besides, loss of tolerance to self-antigens results in pathological conditions which are manifested as autoimmune diseases. Moreover, drugs reducing the antigen presenting capacity of the immune system are therapeutically beneficial in the treatment of rheumatoid arthritis, systemic lupus erythematosus, and multiple sclerosis. SPPL2a (Signal Peptide Peptidase Like 2a), an intramembrane aspartyl protease, engages in the function of B-cells and conventional dendritic cells in mice and humans. Additionally, SPPL2a participates in the intramembrane-cleavage of the membrane-bound N-terminal p8 fragment (NTF) of CD74. Recently, a study from Juraj Velcicky discovered and identified an orally active inhibitor for SPPL2a, SPL-410. SPL-410 represents a highly potent and selective SPPL2a inhibitor.
Strikingly, SPL-410, a hydroxyethylamine based SPPL2a (Signal Peptide Peptidase Like 2a) inhibitor, exhibits an IC50 of 9 nM.
Despite being an attractive target for modulation of the immune system, selective SPPL2a inhibitors are still lacked until now. SPL-707, a novel SPPL2a inhibitor, confirmed that SPPL2a leads to the accumulation of its substrate CD74/p8 and as a consequence to a reduction in the number of B-cells as well as myeloid dendritic cells in mice. In this study, the authors described the hydroxyethylamine based SPPL2a inhibitor SPL-410. SPL-410 15 (compound) inhibited in vivo CD74/p8 fragment processing in mice at 10 mg/kg oral dose.
Further experiments both in vitro and in vivo about the effect of SPL-410 need to be carried out.