ZAP-70 is a 70-kDa ζ-chain CD3 receptor protein tyrosine kinase that operates in the T-lymphocyte signalling pathways. The stimulation of the T-cell receptor triggers the T-lymphocyte signalling pathways. Particularly, ZAP-70 is a critical molecule for transducing T-cell antigen receptor signaling. In addition, ZAP-70 plays an essential role in T-cell signaling. The misregulation of ZAP-70 can lead to immune-related diseases. The overactivation of ZAP-70 can result in autoimmune diseases, while its absence can cause severe combined immune deficiency (SCID). Upon activation of the T-cell antigen receptor, ZAP-70 is recruited to the intracellular ζ chains of the T-cell receptor. The interaction of ZAP-70 with the CD3 ζ chain of the T-cell antigen receptor plays a critical role in T-cell signal transduction. Furthermore, the transduction connects extracellular ligand binding to intracellular signaling events.
Inhibitors of ZAP-70 could potentially function as treatments for autoimmune diseases or organ transplantation. In particular, ZAP-180013 is an inhibitor of the association of ZAP-70 with the T-Cell receptor. In this study, researchers present the design, optimization, and implementation of a screen for inhibitors that would disrupt the interaction between ZAP-70 and the T-cell antigen receptor. Recognition of immunoreceptor tyrosine-based activation motifs (ITAMs) on the T-cell receptor by the tandem SH2 unit of ZAP-70 facilitates T-cell activation in two ways.
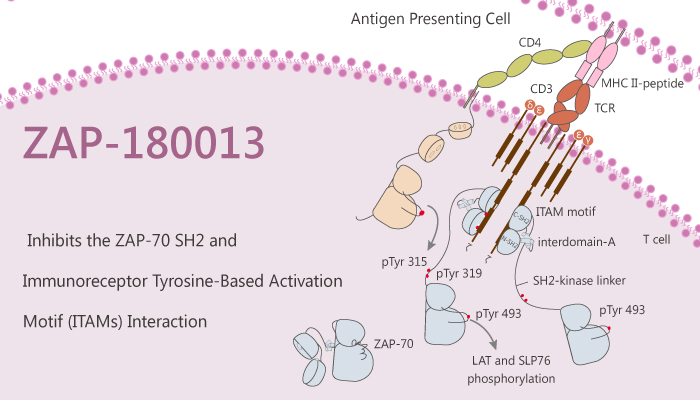
To summarise, blocking ZAP-70 recruitment to phosphorylated ITAMs acts as a means of inhibiting T-cell signaling. Moreover, the over-expression of the isolated tandem SH2 domains of ZAP-70 has a dominant-negative effect. In addition, the effect blocks downstream signaling. As a result, ZAP-180013 may block the function of the ZAP-70 tandem SH2 domains by covalent modification.