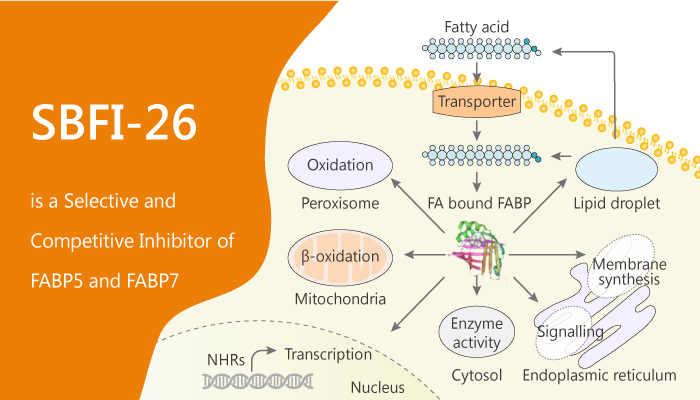
Inflammatory pain is typically treated with non-steroidal anti-inflammatory drugs. However, prolonged use of non-steroidal anti-inflammatory drugs associates with significant side-effects. Fatty acid-binding proteins (FABPs) are a family of small, highly conserved, cytoplasmic proteins that bind long-chain fatty acids and other hydrophobic ligands. FABPs are intracellular lipid chaperones that expressed in cells of the central and peripheral nervous systems. FABPs play roles in fatty acid uptake, transport, and metabolism. Importantly, SBFI-26 is a selective FABP inhibitor.
FABP5 is a cytosolic protein binding with a high affinity to medium and long-chain fatty acids. In particular, FABP5 is a promising target for controlling pain and inflammation. FABP7 binds DHA with the highest affinity among all of the FABPs. Crucially, FABP7 plays a role in cell proliferation and invasion. Particularly, SBFI-26 inhibits the activities of FABP5 and FABP7 with Ki values of 0.9 µM and 0.4 µM, respectively. Especially, SBFI-26 is useful for treating pain and inflammation.
SBFI-26 is the active component of the traditional Chinese medicine Incarvillea Sinensis. In addition, SBFI-26 shows efficient anti-tumor roles in the mouse model. Moreover, SBFI-26 acts as an anti-tumor agent to treat castration-resistant prostate cancer (CRPC). SBFI-26 interferes with the FABP5-PPARγ signaling pathway at the initial stage of the signal transduction by binding competitively to FABP5 to inhibit cellular fatty acid uptake. Furthermore, SB-FI-26 (20 mg/kg, i.p.) produces anti-nociceptive and anti-inflammatory effects in mice. As a result, SBFI-26 produces anti-nociceptive and anti-inflammatory effects in mice.
In summary, SBFI-26 inhibits the activities of FABP5 and FABP7 and produces anti-nociceptive and anti-inflammatory effects.