MyD88 is an adaptor protein. It plays an essential role in the intracellular signaling elicited by IL-1R and several TLRs. MyD88 has a Toll/IL-1R translation initiation region (TIR) domain heterodimerize with the receptor. Besides, MyD88 can recruit downstream signaling molecules such as the serine/threonine kinases, IL-1R-associated kinase 1 (IRAK1) and IRAK4.
ST 2825 is a specific inhibitor of MyD88 dimerization.
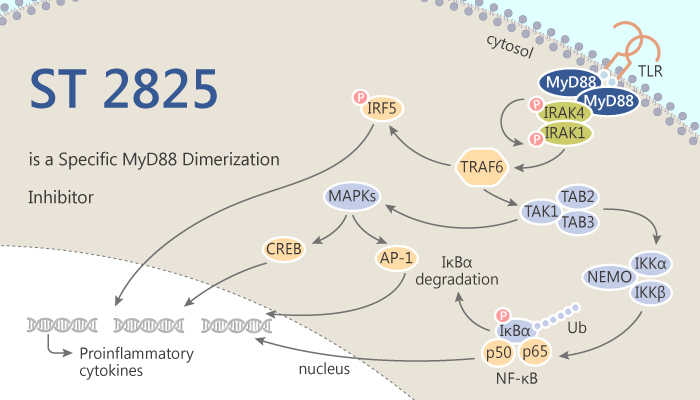
ST 2825 is a synthetic compound that mimics the structure of the heptapeptide in the BB-loop of the MyD88-TIR domain.
In vitro, Firstly, in HEK293T cells, ST2825 specifically inhibits homodimerization of MyD88 TIR domains with 40% inhibition of dimerization at 5 μM and 80% inhibition at 10 μM. Besides, it does not affect the activation of NF-κB induced by poly(I:C). poly(I:C) is a ligand that stimulates the MyD88‐independent TLR3.
Secondly, a full-length MyD88 with its isolated domain is performed in HEK293T cells. ST 2825 at 5 μM only interferes association with the TLR domain, but it does not affect recruitment death (DD) domains. Meanwhile, ST 2825 causes inhibition of IL-1-mediated activation of NF-κB transcriptional activity.
MyD88 dimerization needs to actives its down-stream kinase IRAK1 and IRAK4 activation.
The interaction between MyD88 and IARK is transient. Their derivatives can stabilize this interaction. In HEK293T cells, Flag-MyD88 and Myc-IRAK1 KD or Myc-IRAK4 KD interact strongly.
ST 2825 at 10μM can suppress this interact completely. Besides, it causes a 70% inhibition of MyD88 homodimerization in HEK293T cells.
Lastly, Maria Loiarro et al. determines the ST2825 function in Vivo. They inject C57Bl/6 mice with saline or recombinant murine IL-1β (20 μg/Kg). ST2825 (100-200 mg/Kg ) exerts a significant inhibition of IL-1β-stimulated production of IL-6 by oral administration.
In conclude, ST 2825 blocks IL-1R/TLR signaling by interfering with MyD88 homodimerization and recruitment of IRAK1 and IRAK4 in Vitro. moreover, ST2825 attenuates IL-1β-stimulated IL-6 production in vivo.
Reference
Loiarro M, et al. J Leukoc Biol. 2007 Oct;82(4):801-10.