Irritable bowel syndrome (IBS-C) and chronic constipation are highly prevalent functional gastrointestinal disorders. According to several study datas, IBS-C is characterized by symptoms of chronically recurring abdominal pain, discomfort and bloating in association with symptoms of constipation. In addition, chronic constipation symptoms include infrequent bowel movements, hard stool, straining during defecation, and a feeling of incomplete evacuation. Besides, IBS-C occurs more frequently in women than men. Abdominal discomfort and bloating may also be present. However, for both disorders, current therapeutic options are limited. Hence, we will introduce a guanylate cyclase C (GC-C) agonist-Linaclotide.
Linaclotide is an orally active and selective guanylate cyclase C agonist.
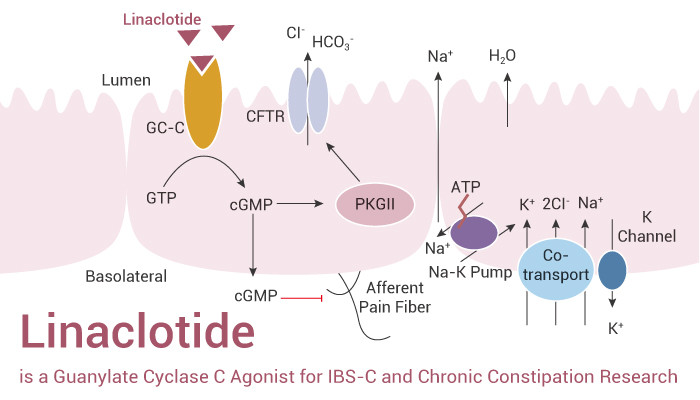
Linaclotide is a 14-amino acid peptide of the guanylin family of cGMP-regulating peptides that also includes the heat-stable microbial ST peptides. Moreover, the transmembrane receptor GC-C expresses on enterocytes along the intestine.
In vitro, Linaclotide (5 µg/mL; 30 min) stimulates fluid secretion by recruiting CFTR to the apical surface of rat small intestine. In addition, Linaclotide (1 µM and 3.27 µM; the 3.27 µM concentration is equivalent to the 5 µg dose of Linaclotide that was used in vivo in the rat jejunum loop) increases phosphorylation of VASP (vasodilator-stimulated phosphoprotein) at Serine 239 in the C2BBe and T84 cells. Besides, Linaclotide (1 µM; 30, 60 min) stimulates cGMP accumulation and short circuit current in human intestinal mucosa.
In vivo, Linaclotide (5 µg in 100 µL KRGH, pH 7.0; i.v.; single) stimulates fluid secretion and cGMP accumulation in intestinal loops. Moreover, Linaclotide (100 µg/kg; p.o.; single) significantly increases the rate of gastrointestinal transit in male wild type and guanylate cyclase C null mice.
All in all, Linaclotide is a promising GC-C agonist for IBS-C and chronic constipation research.
Reference:
[1] Bryant AP, et al. Life Sci. 2010 May 8;86(19-20):760-5.
[2] Ahsan MK, et al. Physiol Rep. 2017 Jun;5(11):e13299.