CD73, known as ecto-5′-nucleotidase (ecto-5′-NT, EC 3.1.3.5) is a glycosyl-phosphatidylinositol (GPI)-linked 70-kDa cell surface enzyme found in most tissues. Targeting CD73 results in favorable antitumor effects in pre-clinical models. Combined treatments of CD73 blockade with other immune-modulating agents (i.e. anti-CTLA-4 mAb or anti-PD1 mAb) is particularly attractive. Extracellular adenosine (ADO) is present in high concentrations in the tumor micro-environment (TME) and suppresses immune function via inhibition of immune cell activation. Intra-tumoral generation of ADO depends on the sequential catabolism of ATP by two ecto-nucleotidases, CD39 (ATP→AMP) and CD73 (AMP→ADO). Inhibition of CD73 eliminates a major, nonredundant, pathway of ADO production in the TME and can reverse ADO-mediated immune suppression.
Lawson et al identified AB-680, a highly potent, reversible and selective inhibitor of CD73.
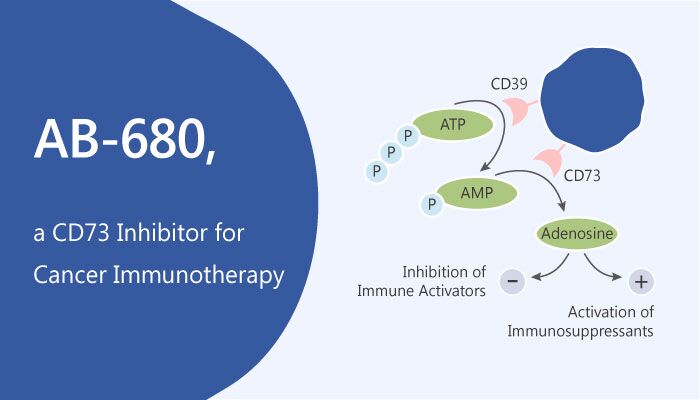
It is more than 10,000-fold selective against related ecto-nucleotidases, such as CD39, NTPDases 2/3/8. This inhibitor is also selective against a large panel of unrelated enzymes, receptors, and ion channels. In contrast to anti-CD73 mAb MEDI9447, it is able to completely inhibit CD73 enzymatic activity on CD8 + T Cells. In the presence of high concentrations of AMP, it robustly restores proliferation of human CD4 + T-cells and reverses AMP mediated inhibition of IFN-γ and IL-2 production. Additionally, AB680 potently restored CD25 expression on human CD8 + T cells. Preliminary toxicology studies show AB680 to be well tolerated following high-dose i.v. infusions in rats. Daily observations (e.g. body weight) did not highlight any difference between dosed animals and vehicle up to 100 mg/kg.
To concluede, AB680 is a highly potent and selective small-molecule inhibitor of CD73 which can mitigate AMP and ADOmediated inhibition of T cell activation. AB680 exhibits a favorable projected human PK profile suitable for parental administration on a schedule consistent with typical mAb dosing cycles. It is expected to enter clinical development in 2018.