Reverse transcriptases (RTs) are enzyme used to generate complementary DNA (cDNA) from an RNA template, a process termed reverse transcription. Reverse transcriptases (RTs) use an RNA template and a short primer complementary to the 3′ end of the RNA to direct the synthesis of the first strand cDNA. Reverse transcriptases are used by viruses such as HIV and hepatitis B to replicate their genomes, by retrotransposon mobile genetic elements to proliferate within the host genome, and by eukaryotic cells to extend the telomeres at the ends of their linear chromosomes.
Nucleoside reverse transcriptase inhibitors (NRTIs) block reverse transcriptase (an HIV enzyme). Non-nucleoside reverse transcriptase inhibitors (NNRTIs) bind to and block HIV reverse transcriptase. HIV uses reverse transcriptase to convert its RNA into DNA (reverse transcription). Blocking reverse transcriptase and reverse transcription prevents HIV from replicating.
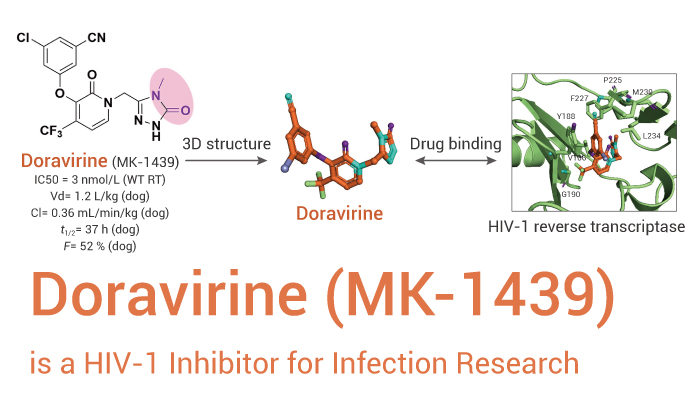
Doravirine (also known as MK-1439) is a highly specific HIV-1 nonnucleoside reverse transcriptase inhibitor.
Doravirine consist of a pyridone core bearing an aryl ether and methyl-triazolone moieties. This compound potently inhibits the wild type and K103N and Y181C reverse transcriptase mutants. Moreover, Doravirine exhibits greater than 10,000-fold selectivity with respect to the cellular DNA polymerases α, β, and γ. Besides, Doravirine shows a selectivity factor of >1,000 versus a broad panel of enzymes, transporters, ion channels, and receptors. In addition, Doravirine does not display cytotoxicity in resting and activated CD4+ T cells, PBMCs, monocytes, and macrophages and in proliferating transformed cell lines, such as MT4, SupT1, and HL60 cell lines. Furthermore, Doravirine has excellent potency in suppressing the replication of wild type, K103N, Y181C, and K103N/Y181C mutant viruses.
In summary, Doravirine is a highly specific HIV-1 NNRTI that potently inhibits the wild type and K103N and Y181C reverse transcriptase mutants.
References:
[1] Lai MT et al. Antimicrob Agents Chemother. 2014;58(3):1652-63.