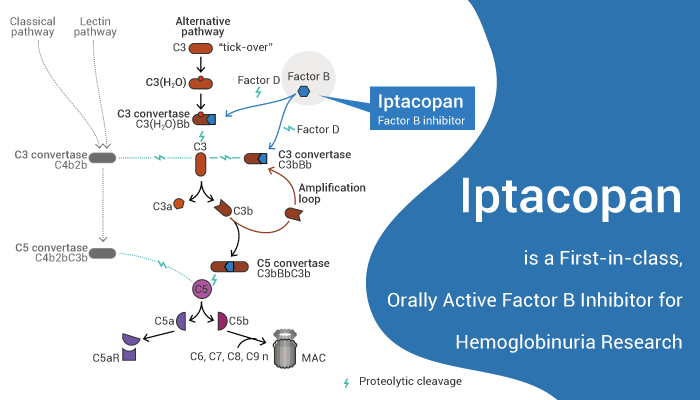
Complement factor B is a protein that in humans is encoded by the CFB gene. Factor B circulates in the blood as a single chain polypeptide. The active subunit Bb is a serine protease that associates with C3b to form the alternative pathway C3 convertase. Bb is involved in the proliferation of preactivated B lymphocytes, while Ba inhibits their proliferation. In addition, the alternative pathway (AP) serves as an amplification loop of complement activation.
Components of the complement system that are limited to the AP include the proteases factor D (FD) and factor B (FB), as well as the γ globulin protein properdin (factor P). FB can bind to C3b or C3(H2O) to generate pro-C3 convertase (C3bB) causing a conformational change that enables the complex to become a substrate for FD.
Iptacopan (also known as LNP023) is a first-in-class, orally active, highly potent and highly selective complement FB inhibitor.
Iptacopan shows direct, reversible, and high-affinity binding to human FB. In addition, Iptacopan exhibits excellent selectivity in a panel of 41 human proteases, including the AP protein factor D. Importantly, Iptacopan is a new drug approved by the FDA for investigational use in paroxysmal nocturnal hemoglobinuria. Besides, Iptacopan demonstrates potent inhibition of alternative complement pathway (AP)-induced membrane attack complex (MAC) formation in 50% human serum. Moreover, Iptacopan (20-180 mg/kg; oral administration) prevents KRN-induced arthritis in mice and is effective upon prophylactic and therapeutic dosing in an experimental model of membranous nephropathy in rats. Furthermore, Iptacopan exhibits moderate half-lives (Wistar Han rats: 3.4 h (30 mg/kg) and beagle dogs: 5.5 h (10 mg/kg) ) following oral administration.
To sum up, Iptacopan is an orally active and highly selective complement FB inhibitor, has the potential for paroxysmal nocturnal hemoglobinuria research.
References:
[1] Anna Schubart, et al. Proc Natl Acad Sci U S A. 2019 Apr 16;116(16):7926-7931.
[2] Nello Mainolfi, et al. J Med Chem. 2020 Jun 11;63(11):5697-5722.