ULK1/2 is an important protein in autophagy for mammalian cells and homologous Atg1 in yeast. Specifically, it is part of the ulk1 complex and is in the early stages of autophagy biosynthesis. Besides, ULK1 is the most basic condition for autophagy, which is activated by multiple upstream signals in the absence of nutrients, and then triggers autophagy. Moreover, ULK1 has many downstream phosphorylation targets to help induce membrane/autophagy. In addition to the function of Atg1 in yeast, ULK1/2 may also play an important role in nerve growth and development. Furthermore, autophagy is a key cellular homeostasis mechanism, and its dysfunction is related to a variety of disease states. It is regulated by the activity of specific kinases, especially UC-51, such as ULK1 and phosphatidylinositol vps34. So these proteins are potential targets for drug development. GW406108X, a potent autophagy inhibitor, shows ATP competitive inhibition against ULK1.
GW406108X, a potent autophagy inhibitor, is a ULK1 and Kif15 (Kinesin-12) inhibitor.
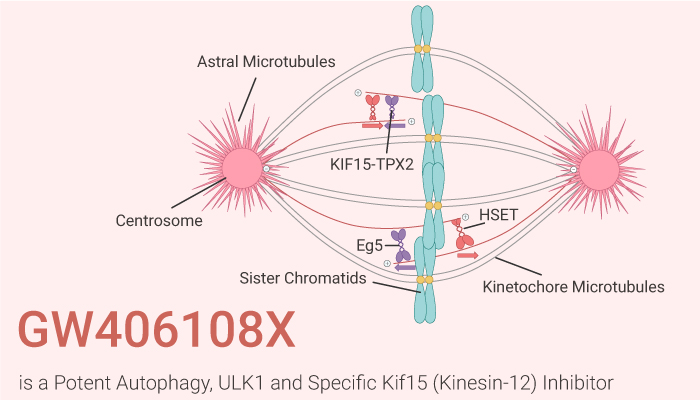
How does GW406108X work on the target? Let’s study it together. In the beginning, GW406108X, a potent autophagy inhibitor, shows ATP competitive inhibition against ULK1 with a pIC50 of 6.37 (427 nM). Meanwhile, GW406108X inhibits ULK1 kinase activity and blocks autophagic flux, without affecting the upstream signaling kinases mTORC1 and AMPK. Nonetheless, GW406108X is a specific Kif15 (Kinesin-12) inhibitor with an IC50 of 0.82 uM in ATPase assays.
In the second place, GW406108X has no affecting mTOR or AMPK activity as shown by monitoring ULK1 pS758 (mTOR site) or pS556 (AMPK site) levels. Particularly, GW406108X inhibits VPS34 and AMPK with pIC50 of 6.34 (457 nM) and 6.38 (417 nM), respectively. Particularly, GW406108X of 5 µΜ significantly reduced the starvation-induced increase in ATG13 phosphorylation. In particular, GW406108X inhibits Kif15-N420 ATPase activity by 76%.
All in all, GW406108X, a potent autophagy inhibitor, is an ATP competitive ULK1 and specific Kif15 (Kinesin-12) inhibitor.
References:
Zachari M, et al. Biochem J. 2020;477(4):801-814.