Kynurenine-3-monooxygenase (KMO) belongs to a family of NADPH dependent flavin monooxygenases. It has an FAD coenzyme, and one gene encodes it. Meanwhile, KMO utilizes either NADPH or NADH, releases NADP+/NAD+ after flavin reduction, and has one Rossmann fold dinucleotide binding domain. Thus, it is a class A flavoprotein monooxygenase (FPMO). In addition, it catalyses the hydroxylation of L-kynurenine (L-Kyn) to 3-hydroxykynurenine (3-HK), using NADPH as a co-substrate. Its expression is largely in the liver, kidney, some monocytic cells, and to a lesser extent in the microglia within the brain.
In animal models, KMO inhibition has shown benefit in neurodegenerative diseases such as Huntington’s and Alzheimer’s. Thus, it is a potential target for acute pancreatitis multiple organ dysfunction syndrome (AP-MODS).
Human KMO is a 56 kDa protein containing a tightly bound FAD cofactor. The C-terminal region attaches to the mitochondrial outer membrane. However, the crystal structure of human KMO is still unknown. On the other hand, the bacterial P. fluorescens enzyme, Pf-KMO, has been most extensively characterized. Therefore, Pf-KMO is a structural surrogate for inhibitor design.
GSK 366 shows potent inhibition at kynurenine-3-monooxygenase (KMO). The IC50 values of 2.3 nM and 0.7 nM for human KMO and Pf-KMO, respectively. Moreover, detailed kinetic analysis for GSK 366 suggests that it true Ki may be around 12 pM. In adition, GSK 366 exhibits a dissociation half-life of the order of 12 hours.
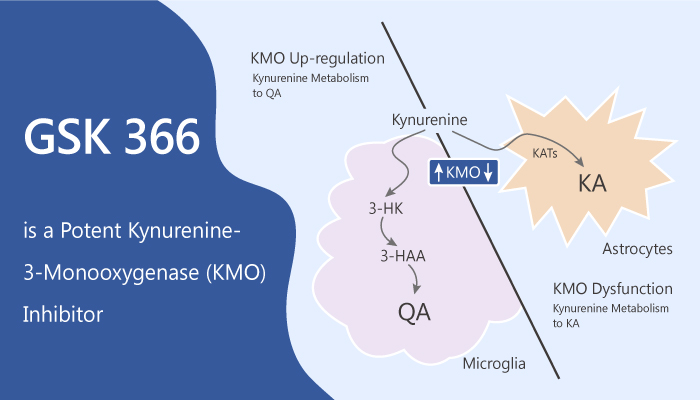
In summary, it is the first reported inhibitor that exploits this flavin motion in class A FPMOs. GSK 366 is suitable candidates for progression to clinical evaluation in acute pancreatitis multiple organ dysfunction syndrome.
Reference: