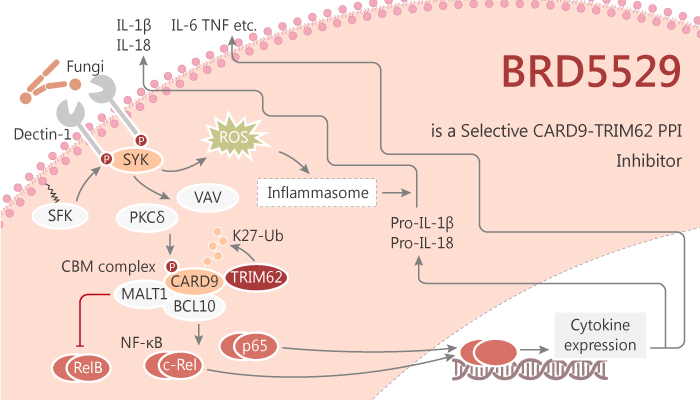
The allele family CARD9 variant relates to increased or protected risk of inflammatory bowel disease (IBD). Moreover, the susceptible variant of CARD9 is related to the increase of NF-κ B-mediated cytokine production. In contrast, the protective variant lacks a functional C-terminal domain and cannot raise E3 ubiquitin ligase trim62. Meanwhile, CARD9 plays a key role in mediating innate immune signals of C-type lectin receptors such as Dectin-1 and mingle. These receptors are responsible for identifying bone marrow cells in fungi and mycobacteria. Although the protective variant CARD9 Δ 11 has a complete N-terminal sequence, it lacks activity and has a major negative impact on CBM signal transmission. E3 ubiquitin ligase trim62 interacted specifically with the WT CARD9 C-terminal domain (CTD) and activated CARD9 by K27 ubiquitination. BRD5529 is a selective CARD9-E3 ubiquitin ligase TRIM62 protein-protein interaction inhibitor.
BRD5529 is a selective CARD9-E3 ubiquitin ligase TRIM62 protein-protein interaction inhibitor with an IC50 of 8.6 μM. Specifically, BRD5529 directly and selectively binds CARD9, disrupts TRIM62 recruitment, inhibits TRIM62-mediated ubiquitinylation and activation of CARD9. Moreover, BRD5529 demonstrates cellular activity and selectivity in CARD9-dependent pathways. Furthermore, BRD5529 significantly reduced IKK phosphorylation in the context of Dectin-1 activation (scleroglucan treatment). But IKK phosphorylation was not affected upon LPS-triggered CARD9-independent TLR4 activation. BRD5529 produced dose-dependent inhibition of TRIM62-mediated CARD9 ubiquitinylation in vitro. Still, BRD5529 functions in a CARD9-specific manner. In addition, BRD5529 attenuated activation of the NF-κB reporter in THP-1 monocytes treated with scleroglucan or whole glucan particles (WGPs). But it had no effect on LPS-driven CARD9-independent NF-κB activation. All in all, BRD5529 is a selective CARD9-E3 ubiquitin ligase TRIM62 protein-protein interaction inhibitor.
References:
Leshchiner ES, et al. Proc Natl Acad Sci U S A. 2017 Oct 24;114(43):11392-11397.