Gamma-aminobutyric acid (GABA) is the chief inhibitory neurotransmitter in the vertebrate central nervous system. It is also a precursor of gamma‐hydroxybutyric acid (GHB). The GABA receptors are a class of receptors that respond to the neurotransmitter GABA. Therefore, they are the chief inhibitory receptors for the central nervous system. There are two classes of GABA receptors: GABAA and GABAB. They play an important role in inhibitory synapses within the central nervous system. GABAA receptors are ligand-gated ion channels (also known as ionotropic receptors); whereas GABAB receptors are G protein-coupled receptors, also called metabotropic receptors.
In ionotropic GABAA receptors, binding of GABA molecules to their binding sites in the extracellular part of the receptor triggers opening of a chloride ion-selective pore. The increased chloride conductance drives the membrane potential towards the reversal potential of the Cl¯ ion, inhibiting the firing of new action potentials. This mechanism is responsible for the sedative effects of GABAA allosteric agonists. In addition, activation of GABA receptors lead to the so-called shunting inhibition. Meanwhile, a slow response to GABA is mediated by GABAB receptors.
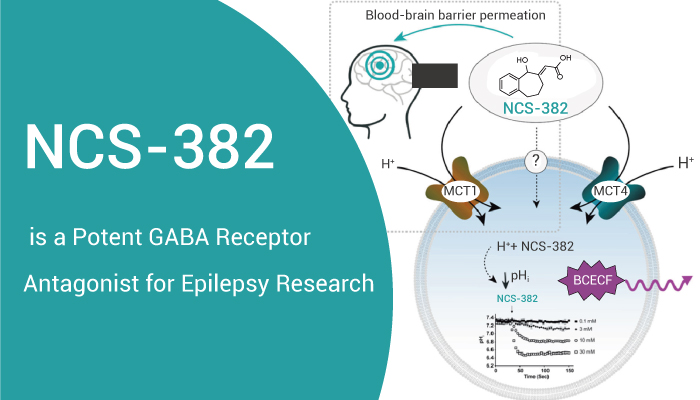
NCS-382 is a potent and moderately selective antagonist of GABA receptor
NCS-382 is a potent GABA receptor antagonist. Therefore, it blocks the effects of GHB in animals. GHB is a minor metabolite of the inhibitory neurotransmitter GABA.GHB can accumulate to significant concentrations in the heritable disorder of GABA degradation, succinic semialdehyde dehydrogenase deficiency (SSADHD). Thus, NCS-382 exhibits anti-sedative and anticonvulsant effects. What’s more, NCS‐382 shows significant efficacy in a murine model of SSADHD, a heritable neurological disorder featuring chronic elevation of GHB that blocks the final step of GABA degradation.
All in all, NCS-382 is a potent and moderately selective antagonist of GABA receptor that exhibits anti-sedative and anticonvulsant effects.
References:
[1] Brohan J, et, al. 2017 Oct;31(10):845-856.
[2] Ainslie GR, et, al. Pharmacol Res Perspect. 2016 Oct 18;4(6):e00265.