HIFs (Hypoxia-inducible factors) are transcription factors that respond to changes in available oxygen in the cellular environment, to be specific, to decreases in oxygen, or hypoxia. The HIF signaling cascade mediates the effects of hypoxia, the state of low oxygen concentration, on the cell. Hypoxia often keeps cells from differentiating. However, hypoxia promotes the formation of blood vessels, and is important for the formation of a vascular system in embryos, and cancer tumors. The hypoxia in wounds also promotes the migration of keratinocytes and the restoration of the epithelium. In general, HIFs are vital to development. In mammals, deletion of the HIF-1 genes results in perinatal death. HIF plays a central role in the regulation of human metabolism. Recently, several drugs that act as selective HIF prolyl-hydroxylase inhibitors have been developed.
Oxygen-dependent hydroxylation of hypoxia-inducible factor (HIF)-α subunits by prolyl hydroxylase domain (PHD) proteins signals their polyubiquitination and proteasomal degradation, and plays a critical role in regulating HIF abundance and oxygen homeostasis. In addition, PHDs may also employ hydroxylase-independent mechanisms to modify HIF activity. Interestingly, while PHDs regulate HIF-α protein stability, PHD2 and PHD3 themselves are subject to feedback upregulation by HIFs. Functionally, different PHD isoforms may differentially contribute to specific pathophysiological processes, including angiogenesis, erythropoiesis, tumorigenesis, and cell growth, differentiation and survival. Because of diverse roles of PHDs in many different processes, loss of PHD expression or function triggers multi-faceted pathophysiological changes as has been shown in mice lacking different PHD isoforms.
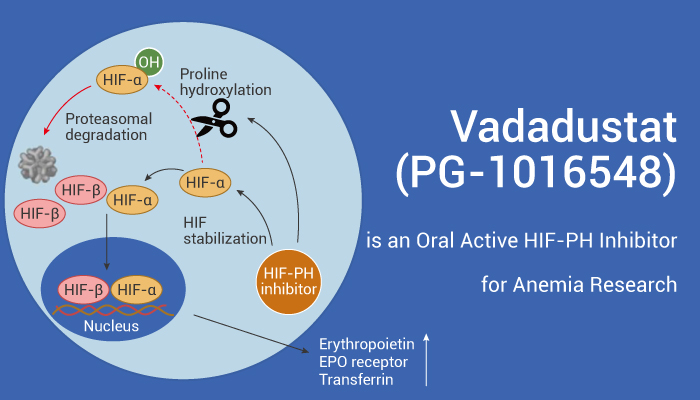
Vadadustat (PG-1016548) is a titratable and orally active HIF-PH inhibitor
Vadadustat is an erythropoiesis-stimulating agent. Besides, Vadadustat induces endogenous erythropoietin synthesis and enhances iron mobilization. Vadadustat increases reticulocytes, plasma EPO, and Hb levels in chronic kidney disease. The increase in plasma EPO levels seen with Vadadustat is comparable in magnitude to that occurring physiologically at moderate altitude and shows a normal diurnal pattern with a return to baseline levels prior to the next dose. Vadadustat improves iron homeostasis by decreasing hepcidin and increasing transferrin levels. Vadadustat exhibits a half-life of approximately 4.5 hours. Overall, Vadadustat has the potential for anemia treatment in chronic kidney disease in vivo.
All in all, Vadadustat is a titratable, orally active HIF-PH inhibitor that has the potential for anemia treatment in chronic kidney disease.
References:
[1] Fong GH, et, al. Cell Death Differ. 2008 Apr;15(4):635-41.
[2] Pergola PE, et, al. Kidney Int. 2016 Nov;90(5):1115-1122.