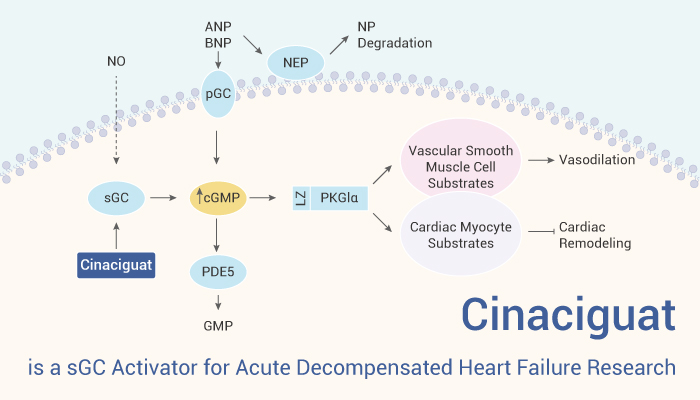
Soluble guanylate cyclase (sGC), the only known nitric oxide (NO) receptor, participates in NO-sGC-cyclic guanosine monophosphate (cGMP) signaling. In the cardiovascular system, sGC is activated by NO, leading to the rapid formation of the second messenger cGMP. cGMP further activates cGMP-dependent protein kinase (PKG), which mediates physiological effects such as vasodilation or platelet aggregation inhibition. Elevated intracellular cGMP levels are cardioprotective. However, the nitro-oxidative stress of NO will directly destroy the structure of sGC, resulting in heme-deficient, NO-insensitive and inactive forms of the enzyme. Here we introduce an activator that is independent of NO and can activate the NO-insensitive form of sGC, Cinaciguat. It enhances impaired cGMP signaling and is a potential inhibitor for the treatment of diabetic cardiomyopathy.
Cinaciquat can prevent structural changes in the diabetic heart and improve heart function.
In in vitro studies, Cinaciguat binds GC with a Kd of 3.2 nM. Cinaciguat (10 μM) significantly enhances intracellular cGMP production. However, there is no dose-dependent effect on cell contraction and calcium transients. Treatment of gastrointestinal smooth muscle strips with Cinaciguat (1-100 nM) showed concentration-dependent relaxation in both WT and apo-sGC mice.
Cinaciguat is orally active. It (10 mg/kg/day, po) does not affect blood glucose levels in diabetic rats but reduces water intake in rats. It also reduces diabetes-associated oxidative stress, prevents diabetes mellitus (DM)-associated alterations in the NO-sGC-cGMP-PKG signaling pathway, and attenuates DM-associated cardiac hypertrophy and apoptosis. Cinaciguat also reduces podocyte damage, cell proliferation and apoptosis in glomeruli of diabetic animals.
In summary, Cinaciguat is an orally available sGC activator with cardioprotective effects.
References:
[1]. Mátyás C, et al. Cardiovasc Diabetol. 2015 Oct 31;14:145.
[2]. Reinke Y, et al. Format: AbstractSend to Eur J Pharmaco.
[3] Czirok S, et al. Sci Rep. 2017 Sep 11;7(1):11218.