Acute Coronary Syndrome (ACS) is a term that describes pathologies related to myocardial ischemia. ACS comprises of unstable angina (UA), Non-ST elevation myocardial infarction (NSTEMI) and ST elevation myocardial infarction (STEMI). Acute management of patients with ACS include antithrombotic therapy. Current medical recommendations of ACS management involve use of dual antiplatelet therapy, typically with aspirin and clopidogrel.
However, newer therapies are being designed and researched to improve outcomes for patients with ACS. Vorapaxar is a novel antiplatelet therapy that inhibits thrombin-mediated platelet aggregation to prevent recurrence of ischemic events. It has been Food and Drug Administration approved for reduction of thrombotic cardiovascular events in patients with a history of MI or peripheral arterial disease with concomitant use of clopidogrel and/or aspirin.
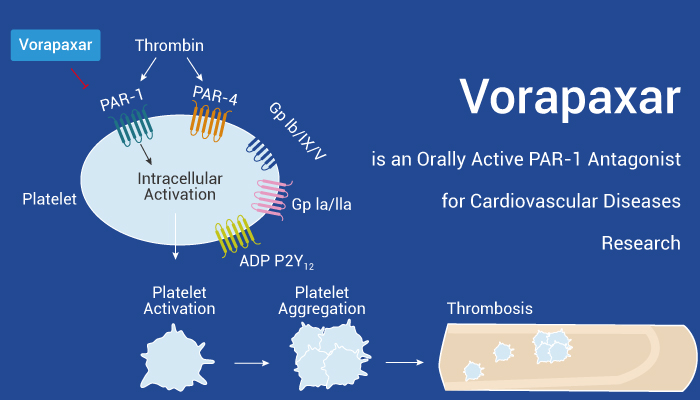
Vorapaxar is an oral medication which inhibits thrombin mediated platelet aggregation.
Vorapaxar (SCH 530348), an antiplatelet agent, is a selective, orally active, and competitive thrombin receptor protease-activated receptor (PAR-1) antagonist (Ki=8.1 nM). Firstly, Vorapaxar inhibits thrombin receptor-activating peptide (TRAP)-induced platelet aggregation in a dose-dependent manner. Secondly, Vorapaxar shows potent inhibition of thrombin-induced platelet aggregation with an IC50 of 47 nM and haTRAP-induced platelet aggregation with an IC50 of 25 nM. What’s more, Vorapaxar inhibits thrombininduced calcium transient in human coronary artery smooth muscle cells (HCASMC) with a Ki of 1.1 nM. Finally, Vorapaxar also inhibits thrombin-stimulated thymidine incorporation in HCASMC with a Ki of 13 nM.
In sum up, Vorapaxar is an oral medication and inhibits thrombin mediated platelet aggregation.
Reference:
[1] Khoufache K, et al. J Clin Invest. 2013 Jan;123(1):206-14.
[2] Kehinde O, et al. J Pharm Bioallied Sci. 2016 Apr-Jun;8(2):98-105.