Hepatitis C virus (HCV) is a positive-strand RNA virus grouped in the genus Hepacivirus within the family Flaviviridae. HCV is classified into at least 6 genotypes (gt), and its error-prone polymerase leads to more than 50 subtypes. The long open reading frame, which encodes the HCV polyprotein, is processed by host and viral proteases and gives rise to three structural proteins (the capsid protein core and envelope glycoproteins E1 and E2) and seven nonstructural (NS) proteins (p7, NS2, NS3, NS4A, NS4B, NS5A, and NS5B).
TMC435 is a SARS-CoV-2 Replication Inhibitor for COVID-19 Research
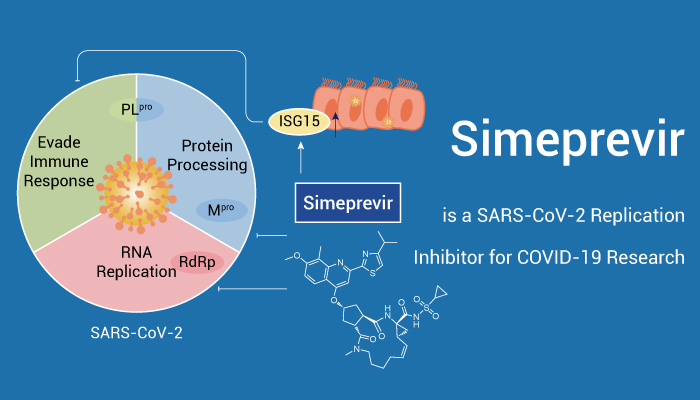
Simeprevir (TMC435; TMC435350) is an oral, potent and highly specific hepatitis C virus (HCV) NS3/4A protease inhibitor with a Ki of 0.36 nM. In addition, Simeprevir inhibits HCV replication with an EC50 of 7.8 nM. Simeprevir also potently suppresses SARS-CoV-2 replication and synergizes with Remdesivir. And it inhibits the main protease (Mpro) and the RNA-dependent RNA polymerase (RdRp) of SARS-CoV-2. And it also modulates host immune responses.
In Vitro, Simeprevir inhibits HCV in a dose-dependent manner in Huh7-Luc cells, with EC50 and EC90 values of 8 nM and 24 nM, respectively. What’s more, Simeprevir inhibits NS3/4A proteases from HCV genotypes 1 to 6 with IC50s of 1/0.9/7/30/1.5/2.2/1.6 nM for 1a/1b/2b/3a/4/5/6, respectively. Morever, it inhibits SARS-CoV-2 in Vero E6 cells with IC50s of 9.6 μM and 5.5 μM for Mpro and RdRp, respectively.
In Vivo, Simeprevir has moderate terminal elimination half-life (t1/2=1.5 h for rat (3 mg/kg, p.o.)). Simeprevir exhibits a medium-slow rate of absorption, well distribution with the high concentration observed in the liver, and a low clearance.
In conclusion, Simeprevir is a SARS-CoV-2 replication inhibitor for COVID-19 research.
Refernce:
[1]. Bioorg Med Chem Lett. 2008 Sep 1;18(17):4853-8.
[2]. Antimicrob Agents Chemother. 2009 Apr;53(4):1377-85. Epub 2009 Jan 26.
[3]. Antimicrob Agents Chemother. 2016 Dec 27;61(1). pii: e01569-16.