Many pathogens have evolved to exploit very particular niches within the host optimally. Candida albicans provides a paradigm for the study of flexible and resilient pathogens. It is the most common cause of fungal infection in humans worldwide. In healthy humans, C. albicans thrives as a harmless commensal of the oral cavity and gastrointestinal tract. Immunocompetent individuals can suffer from recurrent infections, but they typically resolve with minimal morbidity. However, cytochrome bc1 might be the direct target. Cytochrome bc1 is an 11-protein complex. It embeds in the mitochondrial inner membrane that couples the oxidation of ubiquinol to the reduction of cytochrome C. Moreover, the activity of the complex is essential to pump protons from the mitochondrial matrix into the intermembrane space and maintain the proton gradient required for mitochondrial ATP production. In this study, Inz-5 is a fungal-selective mitochondrial cytochrome bc1 inhibitor.
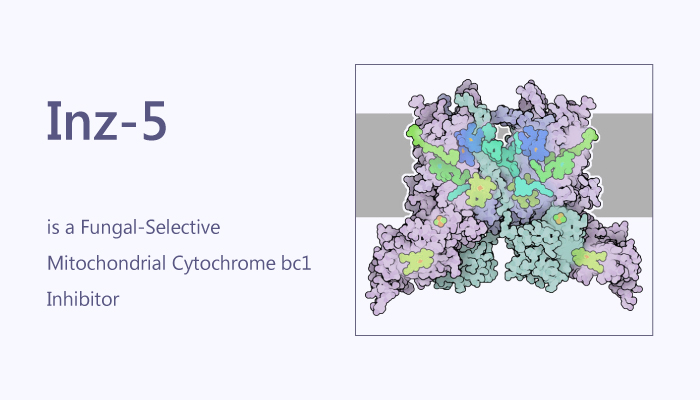
Inz-5 shows activity against Candida proliferation with an IC50 of 0.381 μM. In addition, Inz-5 achieves complete inhibition of C. albicans cytochrome bc1 activity. Inz-5 renders fluconazole fungicidal and reduces the emergence of resistance. Thus, Inz-5 both prevents the de novo emergence of new resistance to fluconazole and transforms fluconazole from a fungistatic compound to a fungicidal one. Furthermore, inhibition of cytochrome bc1 by Inz-5 prevents adaptation to certain carbon sources. It exhibits promising growth inhibition of all three fungi, which is enhanced under respiratory growth conditions.
All in all, Inz-5 can access and occupy a hydrophobic pocket in the inner mitochondrial membrane, but retains sufficient aqueous solubility to achieve adequate levels in the blood poses a formidable challenge. It is an antifungal agent providing a broadly applicable blueprint for similar projects against other medically challenging microbial pathogens.
Reference:
Vincent BM, et al.Cell Chem Biol. 2016 Aug 18;23(8):978-991.