Transglutaminases (TG) belong to a family of structurally and functionally related enzymes. These enzymes catalyse Ca2+-dependent post-translational modifications of proteins. Of these, Transglutaminase 2 (TG2) is an inducible transamidating acyltransferase that catalyzes Ca2+-dependent protein modifications. In addition to being an enzyme, TG2 also serves as a G protein for several seven transmembrane receptors. TG2 also acts as a co-receptor for integrin β1 and β3 integrins distinguishing it from other members of the transglutaminase family. Besides, TG2 expresses in almost all cell types and all cell compartments. Researchers therefore aim to find a highly specific orally active TG2 inhibitor ZED-1227.
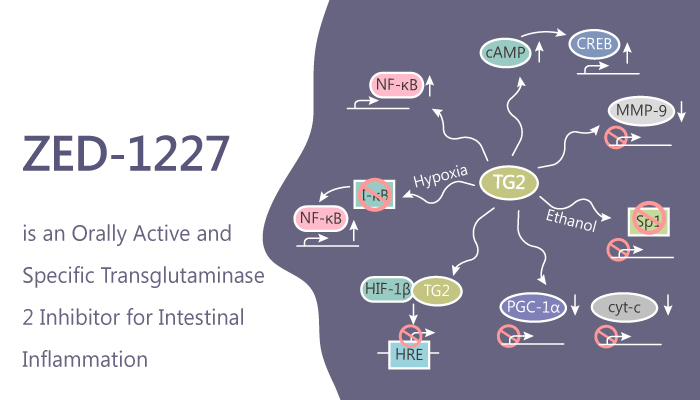
TG2 associates with various human diseases including inflammation, cancer, fibrosis, cardiovascular disease, neurodegenerative diseases, celiac disease. Moreover, TG2 levels especially enhance in the cancer stem cells. TG2 promotes cancer survival, tumor progression and invasion. Furthermore, TG2 can inhibit apoptosis in cancer cells also by directly inhibiting caspase-3 activity via forming a crosslinked multimer, or by upregulating NF-κB activity, which transcribes anti-apoptotic proteins. To illustrate, TG2 activates the NF-κB pathway and promotes inflammation by crosslinking the NF-κB inhibitor IκB-α leading to TNF-α and nitric oxide synthase expression.
In celiac disease, TG2 is a specific target of a conditional autoimmune mechanism driven by exogenous cereal peptides. ZED-1227 can block inflammation-induced TG2 expression and activity, as a preclinical test for a novel pharmacological approach to treat celiac disease. ZED-1227 reduces the activity of intestinal TG2 to normal control levels.
All in all, ZED-1227 is a promising orally available TG2-specific inhibitor, blocking the key step in immunogenic potentiation of gluten peptides in celiac disease.