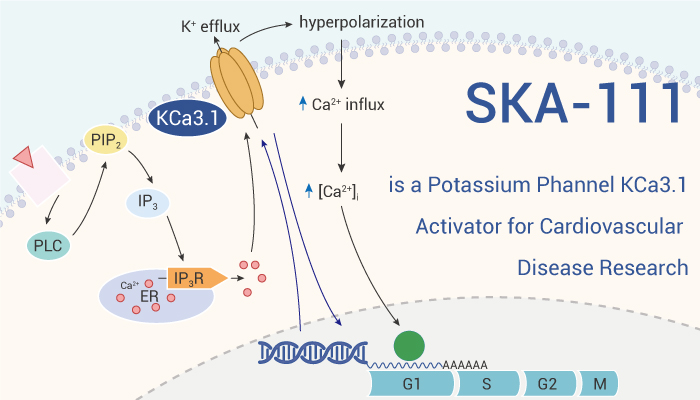
KCa3.1 refers to intermediate-conductance calcium-activated potassium channels, activated via a calmodulin-dependent mechanism. KCa3.1 participates in EDHF-mediated vasculogenesis. Changes in functional ability of KCa3.1 are associated with various cardiovascular diseases and impaired endothelial function. However, KCa3.1 activation provides cardiovascular protection. Beneficial for endothelium-dependent pine relaxation, and vasoconstriction for natural blood vessels. However, the majority of KCa3.1 actiators, can also activate KCa2 channel in the heart and mind, with excessive cardiac relaxation and sedation. This side effects limits KCa3.1 activators to be used for further cardiovascular diseases research. Here, we’ll introduce a highly selective KCa3.1 activator, SKA-111.
SKA-111 acts as a positive gating modulator with hypotensive effect and vasodilator effect.
SKA-111 is a positive KCa3.1 regulator, with 120-fold selectivity against KCa3.1 (EC50=111 nM) over KCa2. Therefore, side effects of KCa2 activation in central nervous system has been avoided. SKA-111 excites KCa3.1 current and induces membrane hyperpolarization in porcine coronary artery (PCA) endothelial cells (PCAEC). It selectively enhances endothelium-dependent PCA relaxation induced by Bradykinin, especially endothelium-derived hyperpolarization (EDH) PCA type relaxation. Even when the NO/frontal gland synthesis is complete, SKA-111 also leads an EDH relaxation in small-caliber PCA. During Langendorff irrigation, SKA-111 (1 μM; 5 min) enhances Bradykinin-induced coronary perfusion pressure (CPP) reduction.
Moreover, SKA-111 (30 mg/kg; i.p.) lowers blood pressure in both normotensive and hypertensive wild-type mouse. Based on pharmacokinetics data from C57Bl/6J mice, indicated a long half-life and a strong brain permeability of SKA-111. SKA-111 shows great potential in cardiovascular disease research.
As mentioned above, SKA-111 is a highly selective KCa3.1 activator, to induce endothelial cell hyperpolarization and coronary arteries relaxation.
References:
[1] Oliván-Viguera A, et.al. . Basic Clin Pharmacol Toxicol. 2016 Aug;119(2):184-92.
[2] Wei AD, et al. Pharmacol Rev. 2005 Dec;57(4):463-72.
[3] Coleman N, et al. Mol Pharmacol. 2014 Sep;86(3):342-57.