Human immunodeficiency virus (HIV) infection causes CD4 T cell depletion and immunodeficiency. Antiretroviral drugs target viral proteins, such as reverse transcriptase, protease, and integrase. Antiretroviral drugs effectively inhibit HIV. However, the low fidelity of the viral reverse transcriptase promotes high levels of viral mutation, frequently leading to HIV drug resistance. Besides, targeting host dependency factors (HDFs) can also inhibit HIV infection. HDFs are cellular proteins that are functionally required for the completion of the viral life cycle. Viruses such as HIV interact with HDFs for essential functions. Targeting HDFs can inhibit viral replication. A dynamic actin cytoskeleton is necessary for viral entry, intracellular migration, and virion release. For HIV-1 infection, during entry, the virus triggers early actin activity by hijacking chemokine coreceptor signaling. This effect activates a host dependency factor, cofilin, and its kinase, the LIM domain kinase (LIMK).
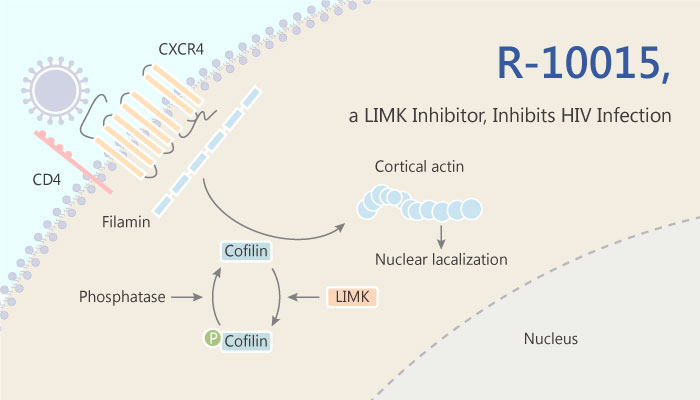
In this study, Fei Yi, et al described the design and discovery of novel classes of small-molecule inhibitors of LIMK for inhibiting HIV infection. Fei Yi, et al identified R-10015 as a lead compound that blocks LIMK activity by binding to the ATP-binding pocket. As a result, researchers selected R-10015 from the potential LIMK inhibitors for further detailed mechanistic studies. R10015 specifically blocks viral DNA synthesis, nuclear migration, and virion release.Researchers performe profiling of R-10015 against a panel of 62 kinases. R-10015 demonstrates reasonably good selectivity (IC50, 38 nM for LIMK1). In addition, R-10015 inhibits multiple viruses, including Zaire ebolavirus (EBOV), Rift Valley fever virus (RVFV), Venezuelan equine encephalitis virus (VEEV), and herpes simplex virus 1 (HSV-1). R-10015 also inhibits viral nuclear migration.
All in all, LIMK inhibitor R-10015 is a new class of broad-spectrum antiviral drugs.