Toll-like receptors (TLRs) are a family of proteins involve in the initial phase of host defence against invading pathogens. All members of this super-family signal inflammation in a similar manner. Especially, TLRs act as primary sensors of microbial products and activate signalling pathways that lead to the induction of immune and inflammatory genes. Most investigations on TLRs have focused on cells of the innate system. Furthermore, most research on the biological implications of TLRs has centred on infections. This is because the patterns recognised by TLRs are principally from pathogens.
Among the TLR family, TLR2 together with TLR1 and TLR6 recognize a wide range of different pathogen-associated molecular patterns (PAMPs) of bacteria, viruses and fungi as well as damage-associated molecular patterns (DAMPs). TLR2 signaling induces antigen presenting cell activation, pro-inflammatory cytokine production and increased expression of co-stimulatory ligand expression. Toll-like receptor 2 ( TLR2) induces early inflammatory responses to pathogen and damage-associated molecular patterns trough heterodimerization with either TLR1 or TLR6. Since overstimulation of TLR2 signaling is linked to several inflammatory and metabolic diseases, TLR2 antagonists may provide therapeutic benefits for the control of inflammatory conditions.
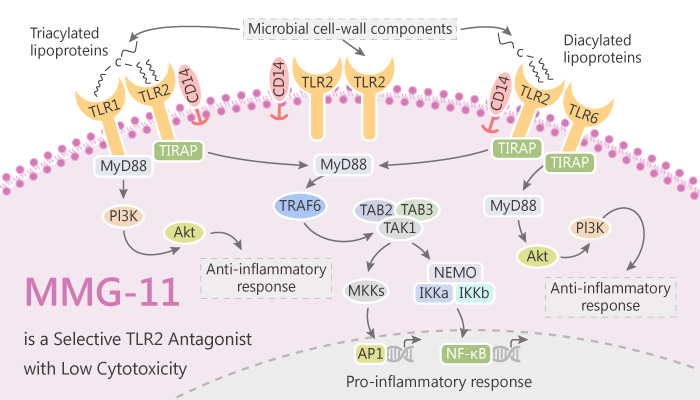
In this study, MariaGrabowski, et al present virtual screening for the identification of novel TLR2 modulators, which combines analyses of known ligand sets with structure-based approaches. The most active compound is MMG-11, which inhibits both TLR2/1 and TLR2/6 signaling. MMG-11 potently inhibit NF-kB activity induced by TLR2 heterodimers. Taken together, researchers demonstrate that MMG-11 is a potent and selective TLR2 antagonist with low cytotoxicity. MMG-11 may be a promising pharmacological tool for the investigation of TLR signaling.